
TUTORIAL 5. LEARNING OUTCOMES By the end of this session the student should be able to: Compare between different titration curves. Select an Indicator. - ppt download

Conversion between Normality, Molarity, Molalityand Mole fraction - Chemistry - Some Basic Concepts of Chemistry - 14513991 | Meritnation.com
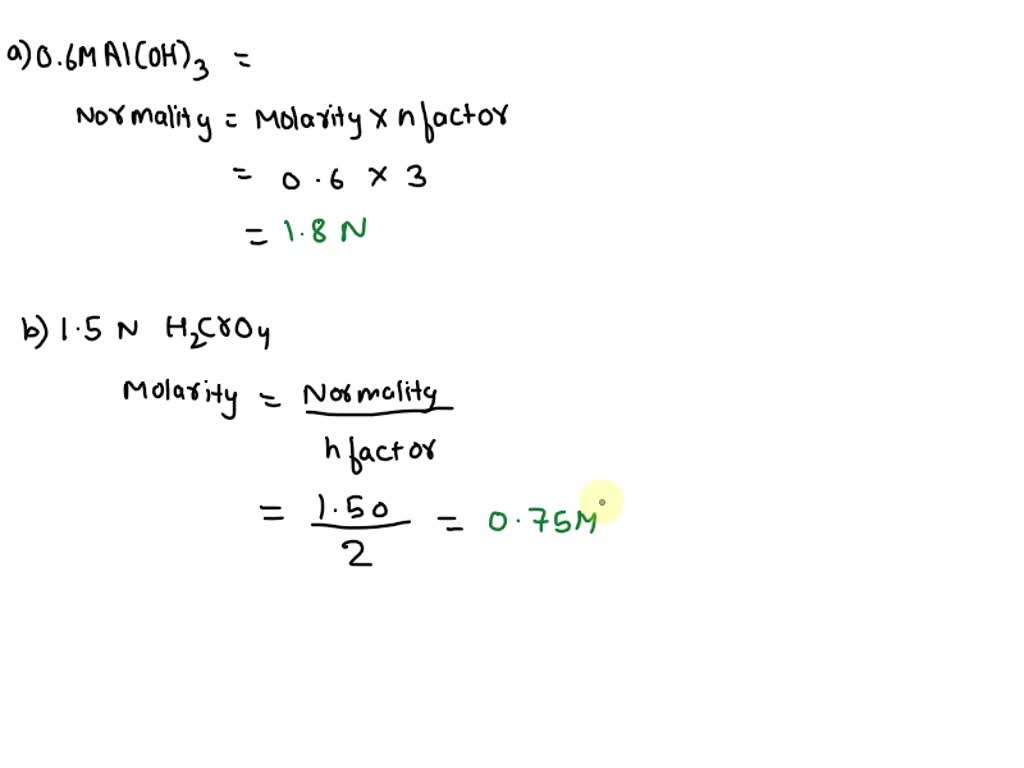
SOLVED: 12. (4 points) Convert Molarity to Normality or vice versa as appropriate 0.66 M Al(OH)3 b. 1.50 N HCrO4 6 M CH3OH d. 0.75 N Ba(OH)2

Calculate the molarity `(M)` and normality `(N)` of a solution of oxalic acid `[(COOH)_(2) . - YouTube


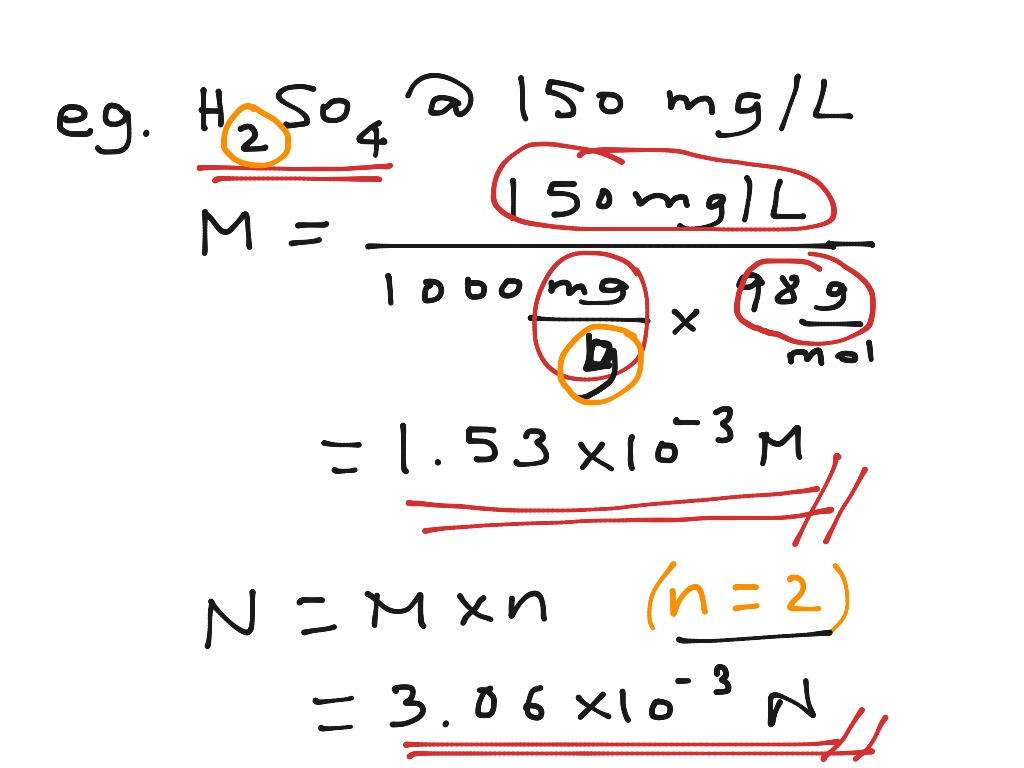


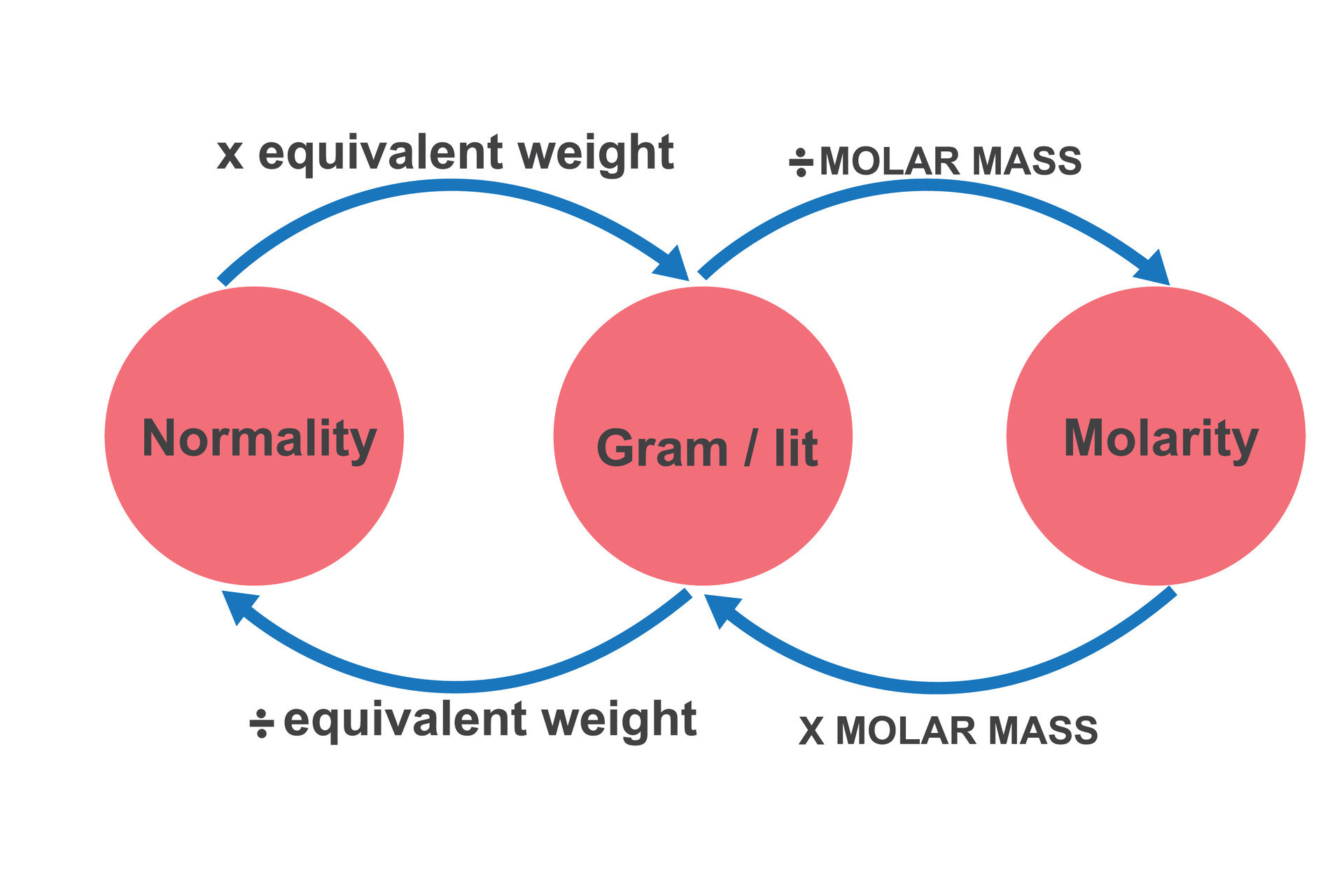







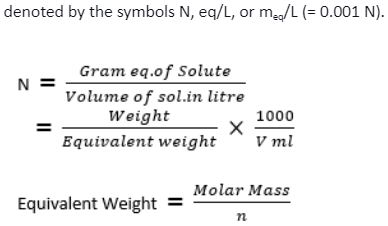


:max_bytes(150000):strip_icc()/how-to-calculate-normality-609580final2-0d5efa5a961f4fa0a7efc780921faee1.png)


