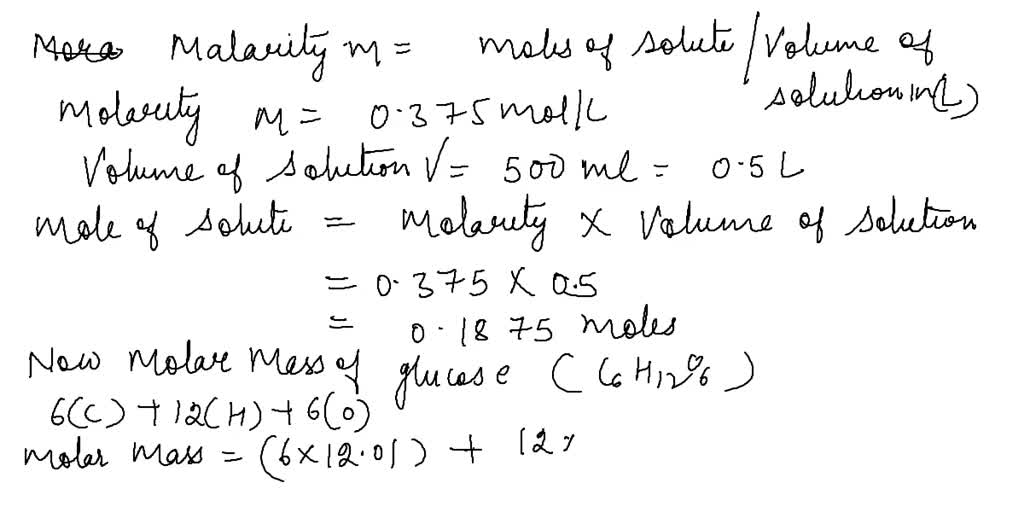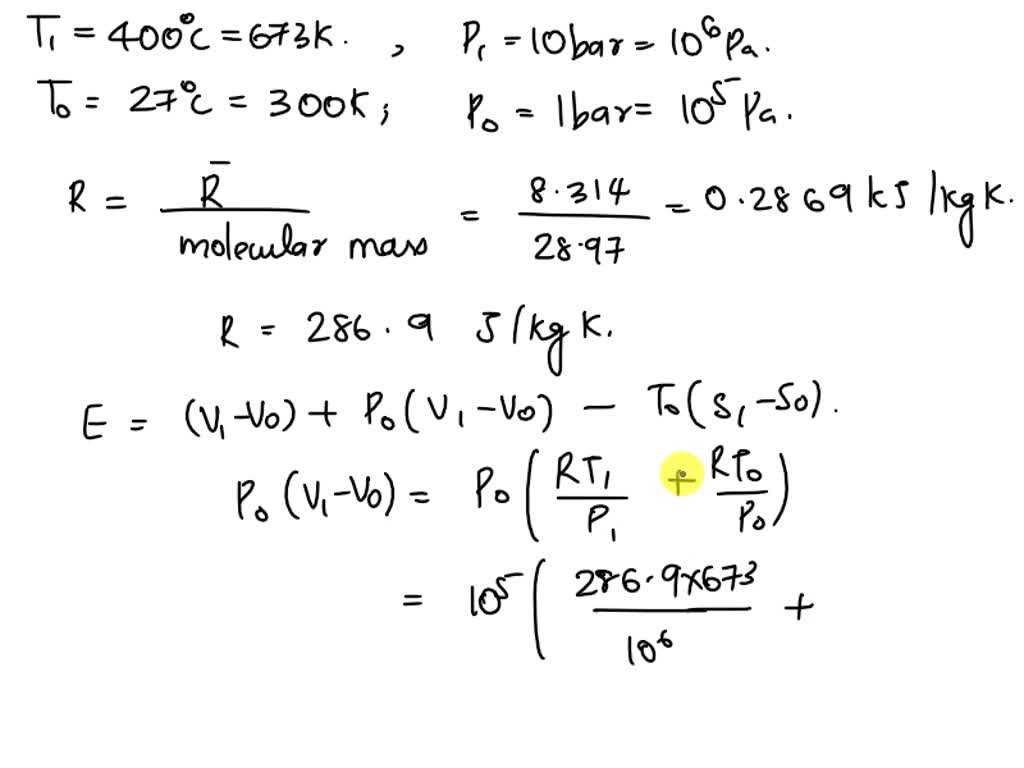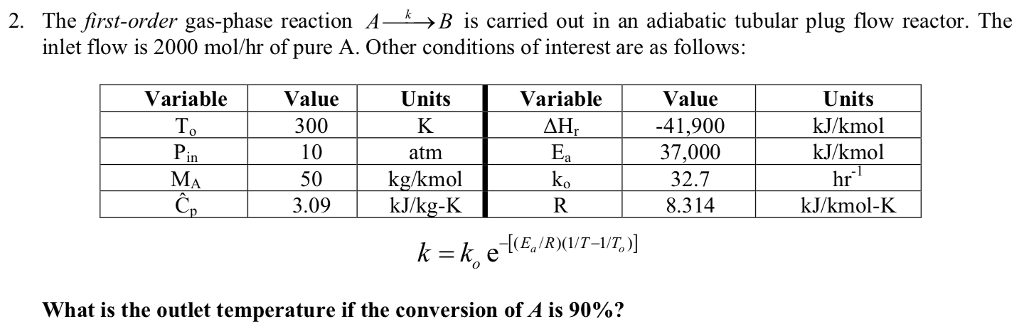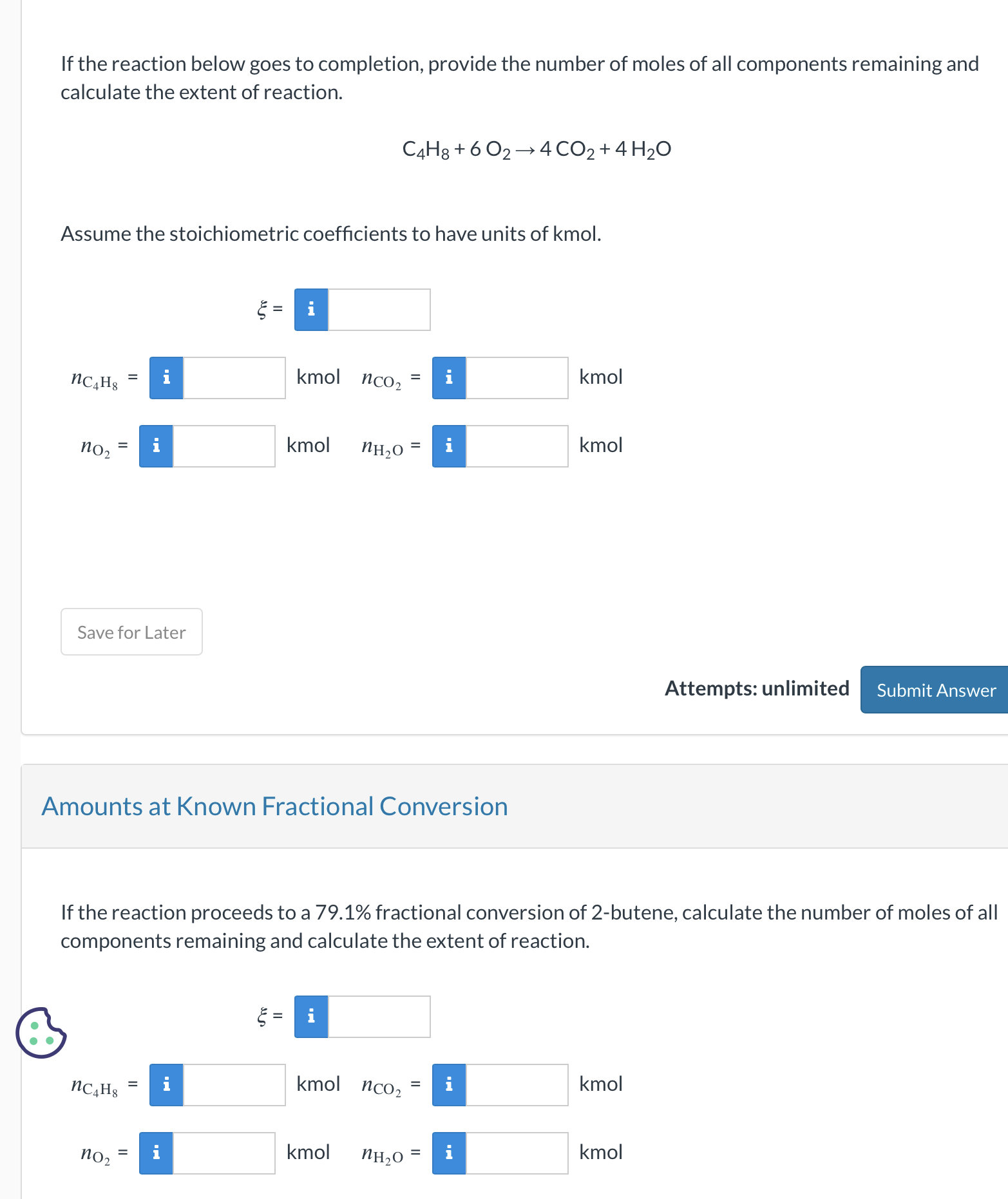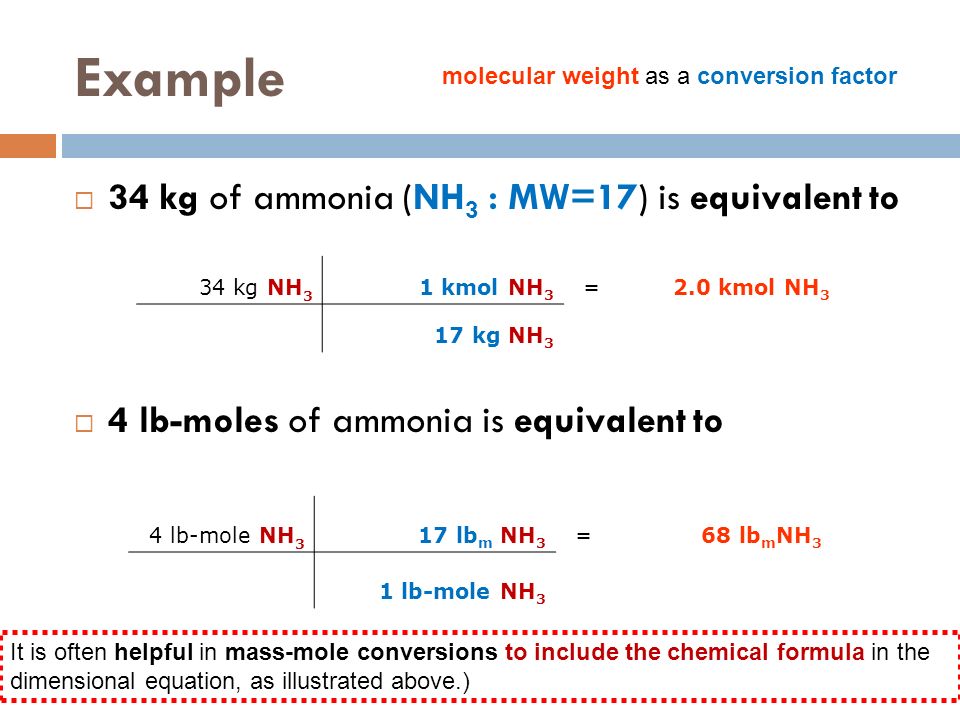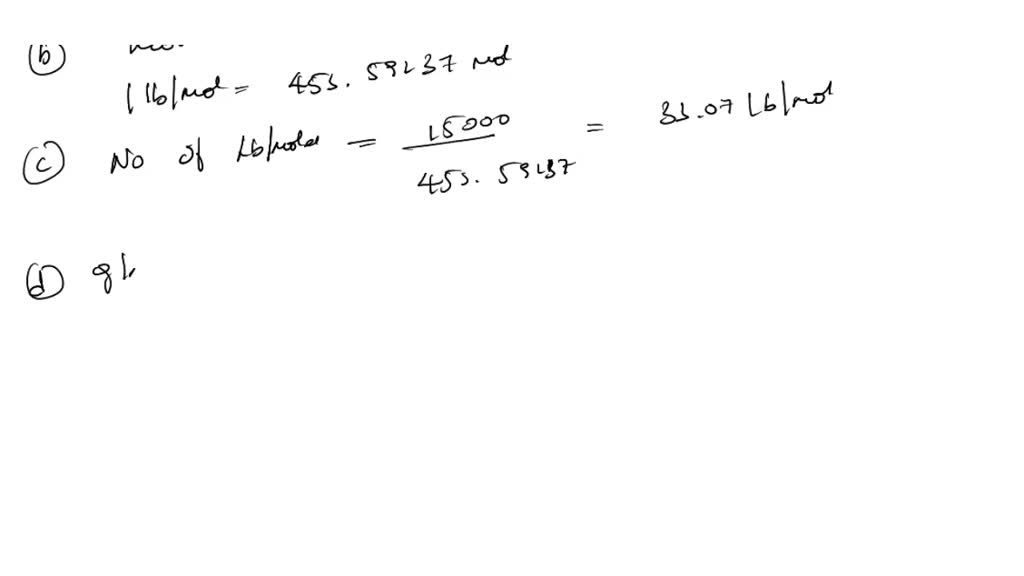
SOLVED: How many of the following are found in 15.0 kmol of benzene (C6H6)? (a) kg CH4; (b) mol C6H6; (c) lb-mole C6H6; (d) mol (g-atom) C; (e) mol H; (f) g
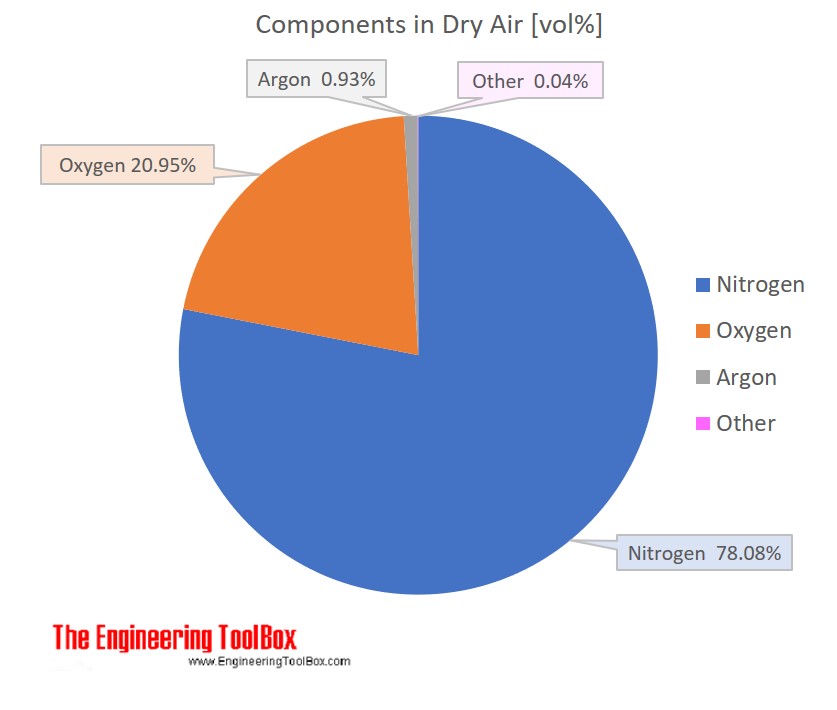
A gas contains 75.0 wt% methane, 10.0% ethane, 5.0% ethylene, and the balance water. (a) | StudySoup

SOLVED: Part 1: A solution with volume V (L) contains n (mol) of a solute A with molecular weight of MA (g A/mol). In terms of V, n, and MA, what is:

SOLVED: The density of air can be calculated using the ideal gas law. If the molecular weight of air is taken to be 28.97 kg/kmol, estimate the air density at 300 K